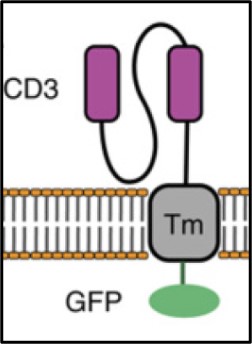
21MB083 TILs: aAPCs that expresses an anti-CD3 scFv transmembrane to determine the cytotoxicity of TILs
An MHC class I and MHC class II negative artificial antigen presenting cell (aAPC) that expresses an anti-CD3 scFv transmembrane construct linked to an intracellular Green Fluorescent Protein (GFP) has been used to develop an assay to determine the cytotoxicity of TILs. This offers a standardized approach to determining TIL cytotoxicity by not focusing on the unknown targets of the TILs that are typically tested using co-culture assays against dissociated autologous tumor cells or biologically relevant HLA-matched cell lines, as there is significant variability in tumor composition and relevant tumor cell targets. The extent of killing can be assayed using flow cytometry. This new T cell potency assay works with all TILsamples, and because it is MHC class I and II negative, works regardless of HLA-typing with minimal allogeneic reactivity, measuring both CD8+ and CD4+ T cell potency.
COMMERCIAL OPPORTUNITY
- The TIL market is attractive as evidenced by the number of companies pursuing TIL therapy including Iovance Biotherapeutics, Instil Bio, Adaptimmune Therapeutics, Achilles Therapeutics, Intima Bioscience, and Turnstone Biologics.
- The solid tumor market is large with about 90% of all cancers being solid tumors, and it is thought that TIL therapies may do better with solid tumors than CAR-T therapies.
- Recently on May 19, 2021, FDA delayed Iovance’s BLA submission for its TIL therapy, Lifileucel and requested additional data on the treatment’s potency assay. This is the second time FDA has delayed Iovance’s TIL product due to concerns over potency assays.
TECHNOLOGY
GFP+ K562 cell expressing an anti-CD3 scFv were co-cultured for 24 hours with a post-REP TIL product. The specific cytotoxicity of the products was measured using a flow cytometry-based assay. K562 aAPC GFP+ cells showed almost 100% cytotoxicity (see example to the right). Another method might use two dyes: one for all target cells and one for the dead target cells in order to determine the percent of dead target cells.
PUBLICATION/PATENT
Provisional patent application filed in November 2021 for Drs. Davila, Beatty and Pilon Thomas
The Innovation Office
InnovationMarketing@Moffitt.org
(813) 745-6828

