19MA010 Med Device: Drug Infusion 'The Snowflake' Device for Treatment of Brain Cancer
Snowflake infusion device for drug delivery used in treatment of glioblastoma. After surgery in the brain, a device is needed to provide drug equally to an irregular region of the brain and to reduce backflow and leakage. Providing localized drug to the tissue can often limit the extent of de-bulking needed, helping to preserve affected organ function. This is critical when the neoplastic tissue is in the brain. The solution, Snowflake, is a catheter system with multiple tips from a central hub connected to a pump to deliver drug into the tumor resection cavity thereby offering more effective drug coverage in the cavity to treat infiltrating tumors like glioblastoma.
COMMERCIAL OPPORTUNITY
- The American Cancer Society estimates that in 2019 there will be 23,820 patients who develop brain cancer and 17,760 brain cancer patients will die.
- Drugs such as Topotecan may benefit from infusion using this device. Topotecan is a topoisomerase I inhibitor that causes single-stranded DNA breaks during DNA replication. Topotecan can target mitotically active cells in a quiescent environment. Systemic application of topotecan has been limited by poor blood brain barrier penetration, and Phase II studies showed no efficacy with severe side effects. Preclinical rat studies using convection-enhanced delivery of topotecan showed a significant survival advantage. A Phase Ib dose-escalation study in patients with recurrent malignant gliomas demonstrated radiographic tumor regression in 69% of patients at nontoxic concentrations (Neurotherapeutics 2017 Apr; 14(2): 358–371).
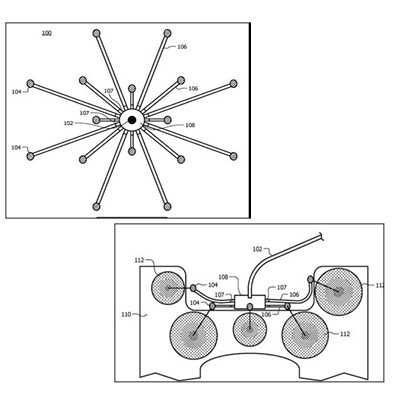
TECHNOLOGY
Snowflake infusion device comprises of a number of delivery tubes from a central hub with varying lengths and internal diameters so that when the device is implanted there is equal impedance to flow through each of the attached delivery tubes resulting in equal flow out of the delivery tubes to regions of interest in the patient's brain. The device allows clinicians to customize the flowrate in different delivery tips from a single input hub and deliver the drugs as required in irregular regions of interest in brain and reduce backflow and leakage.
PUBLICATION/PATENT
PCT patent application filed on May 13, 2020 for Dr. Michael Vogelbaum.
The Innovation Office
InnovationMarketing@Moffitt.org
(813) 745-6828

