Can a Personalized Treatment Stop Bladder Cancer From Coming Back?
An experimental cancer immunotherapy designed to train the immune system to better recognize bladder tumors is being tested in a clinical trial with standard treatment used for high-risk, early-stage bladder cancer.
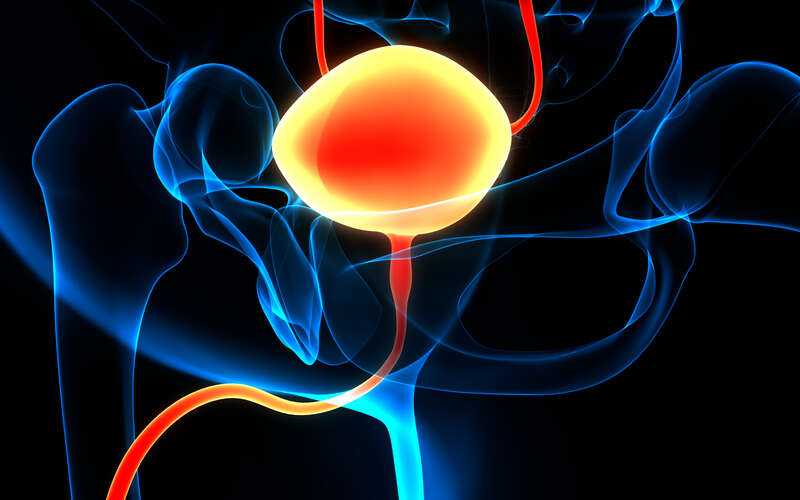
The study focuses on patients with high-risk non-muscle invasive bladder cancer (NMIBC), a form of the disease that remains confined to the inner lining of the bladder but carries a significant risk of returning after treatment.
Standard care includes surgical removal of tumors followed by Bacillus Calmette-Guerin, or BCG, an immunotherapy delivered into the bladder to lower the chance of recurrence. While BCG has been used for decades and works for many patients, recurrence rates remain high.
The phase 2 clinical trial is evaluating whether adding a personalized mRNA treatment, intismeran autogene (V940), to BCG helps patients stay cancer-free longer than BCG alone.
A Personalized Approach
V940 is created individually for each patient. After surgery, a tumor sample is analyzed to identify mutations specific to that cancer. The treatment is then designed to recognize and attack cells carrying those mutations.
Similar personalized mRNA treatments have shown encouraging results in other cancers, including melanoma.
Roger Li, MD, a urologic oncologist at Moffitt Cancer Center, presented the trial design in the Trials-in-Progress session at the 2026 American Society of Clinical Oncology Genitourinary Cancer Symposium.

Roger Li, MD
“In many different cancer types, this personalized therapy is combined with immune checkpoint blockade in the higher disease stages,” Li explained. “This trial is unique in that V940 is added to BCG, a different form of immunotherapy, to treat disease at a much earlier stage to prevent the tumor from coming back.”
How the Study Works
In the trial, nearly 300 patients are being randomly assigned to receive either BCG alone or BCG in combination with V940. Investigators will compare how long patients remain free from recurrence or progression and monitor safety.
A smaller group of patients who are unable or unwilling to receive BCG will receive V940 alone.
“I think this is one step closer to finding out if an immune therapy combined with BCG is going to be clinically significant in preventing disease recurrence or progression,” Li said. “It’s a very novel way to approach the disease and understanding its molecular makeup, how to activate a patient’s personal immune system and use that in combination with standard-of-care to improve outcomes.”
The study is ongoing and continues to enroll patients.